The battery which uses sponge lead and lead peroxide for the conversion of the chemical energy into electrical power, such type of battery is called a lead acid battery. The lead acid battery is most commonly used in the power stations and substations because it has higher cell voltage and lower cost.
Construction of Lead Acid Battery
The various parts of the lead acid battery are shown below. The container and the plates are the main part of the lead acid battery. The container stores chemical energy which is converted into electrical energy by the help of the plates.
1. Container
The container of the lead acid battery is made of glass, lead lined wood, ebonite, the hard rubber of bituminous compound, ceramic materials or moulded plastics and are seated at the top to avoid the discharge of electrolyte. At the bottom of the container, there are four ribs, on two of them rest the positive plate and the others support the negative plates.
The prism serves as the support for the plates and at the same time protect them from a short-circuit.The material of which the battery containers are made should be resistant to sulfuric acid, should not deform or porous, or contain impurities which damage the electrolyte.
2. Plate
The plate of the lead-acid cell is of diverse design and they all consist some form of a grid which is made up of lead and the active material. The grid is essential for conducting the electric current and for distributing the current equally on the active material. If the current is not uniformly distributed, then the active material will loosen and fall out.
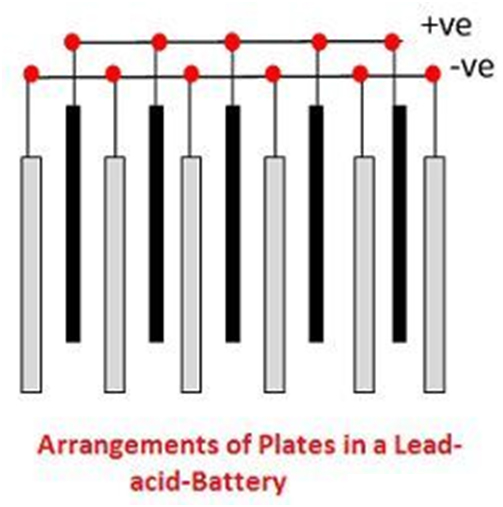
The grids are made up of an alloy of lead and antimony. These are usually made with the transverse rib that crosses the places at a right angle or diagonally. The grid for the positive and negative plates are of the same design, but the grids for the negative plates are made lighter because they are not as essential for the uniform conduction of the current.
The plates of the battery are of two types. They are the formed plates or plante plates and pasted or faure plates.
Plante’s plates are used largely for stationary batteries as these are heavier in weight and more costly than the pasted plates. But the plates are more durable and less liable to lose active material by rapid charging and discharging. The plantes plate has low capacity weight-ratio.
Faure process is much suitable for manufacturing of negative plates rather than positive plates. The negative active material is quite tough, and it undergoes a comparatively low change from charging and discharging.
3. Active Material
The material in a cell which takes active participation in a chemical reaction (absorption or evolution of electrical energy) during charging or discharging is called the active material of the cell. The active elements of the lead acid are
The lead peroxide and sponge lead, which form the negative and positive active materials have the little mechanical strength and therefore can be used alone.
4. Separators
The separators are thin sheets of non-conducting material made up of chemically treated leadwood, porous rubbers, or mats of glass fibre and are placed between the positive and negative to insulate them from each other. Separators are grooved vertically on one side and are smooth on the other side.
5. Battery Terminals
A battery has two terminals the positive and the negative. The positive terminal with a diameter of 17.5 mm at the top is slightly larger than the negative terminal which is 16 mm in diameter.
When the sulfuric acid dissolves, its molecules break up into positive hydrogen ions (2H+) and sulphate negative ions (SO4—) and move freely. If the two electrodes are immersed in solutions and connected to DC supply then the hydrogen ions being positively charged and moved towards the electrodes and connected to the negative terminal of the supply. The SO4— ions being negatively charged moved towards the electrodes connected to the positive terminal of the supply main (i.e., anode).
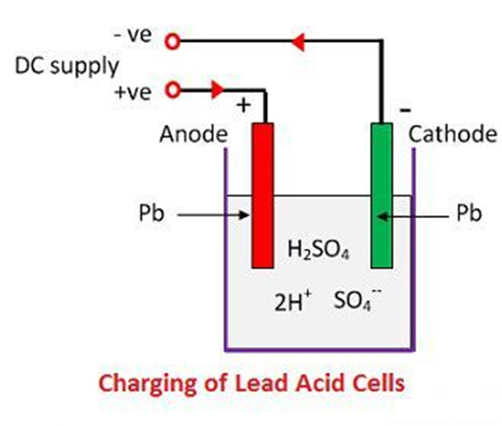
Each hydrogen ion takes one electron from the cathode, and each sulphates ions takes the two negative ions from the anodes and react with water and form sulfuric and hydrogen acid.
The oxygen, which produced from the above equation react with lead oxide and form lead peroxide (PbO2.) Thus, during charging the lead cathode remain as lead, but lead anode gets converted into lead peroxide, chocolate in colour.
If the DC source of supply is disconnected and if the voltmeter connects between the electrodes, it will show the potential difference between them. If wire connects the electrodes, then current will flow from the positive plate to the negative plate through external circuit i.e. the cell is capable of supplying electrical energy.
Chemical Action During Discharging
When the cell is full discharge, then the anode is of lead peroxide (PbO2) and a cathode is of metallic sponge lead (Pb). When the electrodes are connected through a resistance, the cell discharge and electrons flow in a direction opposite to that during charging.
The hydrogen ions move to the anode and reaching the anodes receive one electron from the anode and become hydrogen atom. The hydrogen atom comes in contacts with a PbO2, so it attacks and forms lead sulphate (PbSO4), whitish in colour and water according to the chemical equation.
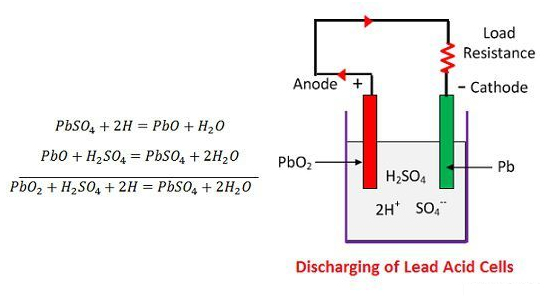
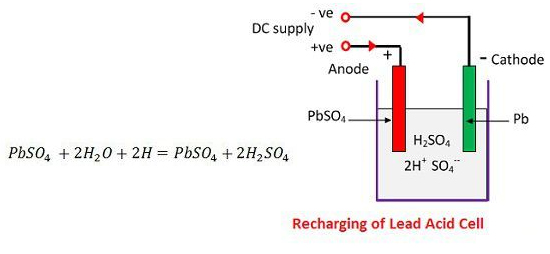
-SO4— ion moves to the anode, gives up its two additional electrons becomes radical SO4, react with the lead sulphate anode and form leads peroxide and lead sulphuric acid according to the chemical equation.
The charging and discharging are represented by a single reversible equation given below.
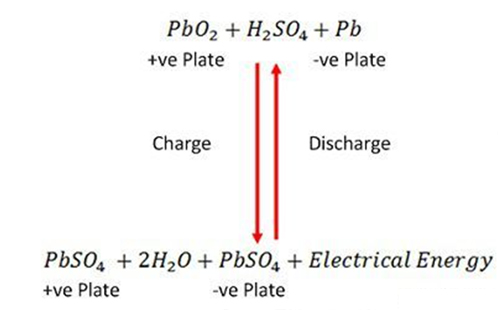
The equation should read downward for discharge and upward for recharge.
Flooded batteries come in the widest variety of shapes and sizes due to their widespread usage in a multitude of industries and applications. They’re the most commonly used batteries on the reserve power (standby) market today.
Flooded batteries are very reliable and robust against harsh environments. For this reason, they are often preferred by majority of larger reserve power applications.
Flooded batteries do not recombine the hydrogen and oxygen gases to water internally as seen with VRLA batteries; instead, these gases are vented externally. Thus, ventilation must be considered and properly sized to remove the hydrogen gases from the battery area.
Flooded batteries require more maintenance than VRLA batteries. For example, distilled or deionized water has to be routinely replenished to make up for the lost moisture (hydrogen and oxygen).
Tubular batteries are cast from low antimony lead alloy, with selenium, copper, tin, and arsenic, to protect the lead support from anodic corrosion.
Why tubular is better: The secrets are the multi-tube bag gauntlet and increased surface area of the positive plate.
SDS Advanced multi-tube bag gauntlets are constructed of 100% polyester high-tenacity, multifilament yarns that are impregnated with an acrylic resin system and fixed crosswise to the tubes, while spun yarn is used lengthwise.
Tubular battery technology seals the active material in polyester tubes called gauntlets,instead of pasting it on the surface of the plate. This has a wonderful result, of no shedding or corrosion, ensuring long battery life. As tubular batteries have desired toughness and durability, It can operate at extreme temperatures, and are used in high cyclic applications involving frequent and prolonged power outages.
We take extra care in the composition of the alloy in the positive support structure, as well as in the way that the main back bone of the plates, called the SPINE, is cast, this ensures the long life.
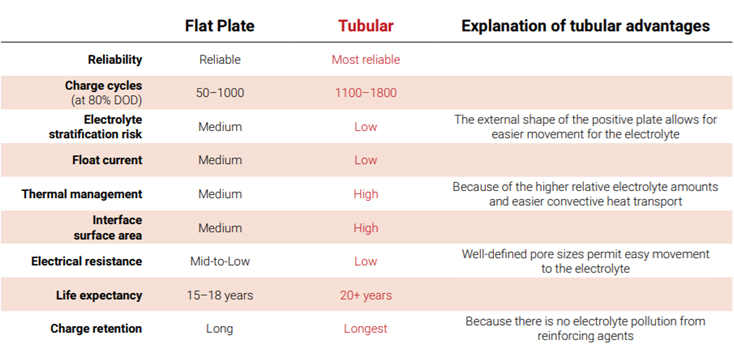
Due to increased positive plate surface area, tubular batteries have 20% more electrical capacity than flat plate batteries of comparable size and weight. With less positive plate shedding, tubular batteriesalso provide up to a 30% longer service life than flat plate batteries. In addition, battery engineers inEurope attest that tubular cells are more widely used because they deliver energy at a faster rate.
Other Stationary Tubular Advantages Doubters of tubular construction might argue that flat plate batteries — due to their simpler construction — are generally less expensive to make and maintain. However, tubular batteries have been shown to have both a longer life and supply more power faster in an equivalent size, while still being produced at a competitive cost.
Perhaps most importantly to stationary applications, the tubular positive grid does not require horizontal bars, which virtually eliminates positive plate growth and therefore post seal leaks and jar cracking. As a result, in applications which require a long service life, tubular plate batteries provide the best and most reliable power for the money spent
Valve-Regulated Lead-Acid or VRLA, including Gel and AGM (Absorbed Glass Mat) battery designs, can be substituted in virtually any flooded lead-acid battery application (in conjunction with well-regulated charging). Their unique features and benefits deliver an ideal solution for many applications where traditional flooded batteries would not deliver the best results.
A VRLA battery utilizes a one-way, pressure-relief valve system to achieve a “recombinant” technology. This means that the oxygen normally produced on the positive plate is absorbed by the negative plate. This suppresses the production of hydrogen at the negative plate. Water (H2O) is produced instead, retaining the moisture within the battery. It never needs watering, and should never be opened as this would expose the battery to excess oxygen from the air. In addition to damaging the battery, opening it also voids the warranty
The difference between VRLA and traditional flooded batteries Flooded electrolyte batteries do not have special one-way, pressure-relief valves, as they do not work on the recombination principle. Instead, flooded designs utilize a vent to allow gas to escape. They contain liquid electrolyte that can spill and cause corrosion if tipped or punctured. They should not be used near sensitive electronic equipment. They can only be installed “upright.” Flooded batteries lose capacity and become permanently damaged if: n Left in a discharged condition for any length of time (due to sulfation). This is especially true of designs that require water maintenance. n Continually over-discharged (due to active material shedding). This is especially true of automotive starting types.
VRLA technology encompasses both gelled electrolyte or gel batteries and absorbed glass mat or AGM batteries. Both types are regulated by special one-way, pressure-relief valves and have significant advantages over flooded lead-acid products. AGM (Absorbed Glass Mat) batteries The electrolyte in AGM batteries is completely absorbed in separators consisting of matted glass fibers. This causes them to be spillproof, meaning they don’t leak acid like a flooded design if tipped on their side. The glass mats in AGM batteries are wrapped around the positive plate, which helps prevent damage from vibration and extend cycling. The battery’s groups are packed tightly in the case partitions also protecting its power producing components. AGM battery designs can have over twice the cycle life of a conventional flooded product in the right application.
Our Pure Lead plated products are valve regulated lead-acid (VRLA) batteries made with absorbed glass mat (AGM) construction.
VRLA-AGM type is a recombinant battery. This means that the oxygen evolved at the positive plates will recombine with the hydrogen generated at negative plates creating water and preventing water loss. Absorbed glass mat (AGM) batteries absorb the electrolyte in a fiber-glass mat separator.
An integrated pressure vent aids oxygen recombination and reduces water loss. A porous fiber glass material is used to separate the plates, retain electrolyte at the surface of the electrodes for electron transfer, and allow oxygen migration. High grade acid is used in conjunction with high purity virgin lead for the grids, active materials, and current collectors. The plates are thin which allows more plates per available volume relative to traditional lead-acid batteries.
The electrode reactions in all lead acid batteries including VRLA battery is basically identical. As the battery is discharged, the lead dioxide positive active material and spongy lead negative active material both react with the sulphuric acid electrolyte to form lead sulphate and water. During charge, this process is reversed. The coulombic efficiency of the charging process is less than 100% on reaching final stage of charging or under over charge conditions, the charging energy is consumed for electrolytic decomposition of water and the positive plates generate oxygen gas and the negative plates generate hydrogen gas. Under typical charging conditions, oxygen at the positive plate occurs before hydrogen evolution at the negative. This feature is utilized in the design of VRLA batteries. In flooded cells, the oxygen gas evolved at the positive plate bubbles upwards through the electrolyte and is released through the vents. In VRLA batteries the oxygen gas evolved at the positive instead of bubbling upwards is transported in the gas phase through the separator medium to the negative plate.
The separator is a highly absorbent glass mat type with very high porosity designed to have pore volume in excess of the electrolyte volume (starved electrolyte design), due to which the oxygen gas finds an unimpeded path to the negative plate. The oxygen gas gets reduced by reaction with the spongy lead at the negative plate turning part of it into a partially discharged condition, thereby effectively suppressing the hydrogen gas evolution at the negative plate. This is known as the oxygen recombination principle. The part of negative plate which was partially discharged is then reverted to original spongy lead by subsequent charging. Thus a negative plate keeps equilibrium between the amount which turns into spongy lead by charging and the amount of spongy lead which turns into lead sulphate by absorbing the Available Range: 12V 6.5AH to 12V 200AH

The technology behind our gel battery has the sulfuric acid electrolyte mixed with silica material to form an immobilized gel. VRLA - Gel batteries are recombinant type and do not require water addition, and emit less gas than VRLA type lead-acid batteries. Gel batteries reduce the electrolyte evaporation and spillage that are common to the VRLA battery. Gel batteries are considered to have greater resistance to extreme service temperatures.
Although Gel batteries belong to a VRLA category, its construction is similar to VRLA (flooded) batteries except for the gelled electrolyte. A Gel battery’s separator material is generally not an Absorbent Glass Mat, but microporous plastic material.
Our Solar Gel batteries are maintenance-free batteries with a special gel electrolyte made by mixing acid electrolyte with pyrogenic silica. The design uses pasted plate grids with a large surface area with grids casted from a special Lead-Calcium-Tin-Silver alloy that provides high corrosion resistance, and a special micro porous synthetic separator.
Advantages
Dry Charged Batteries. For Dry Charged batteries we provide “Hybrid Alloy technology”. Hybrid Alloy technology Batteries have very good Cycling ability and are also low maintenance. By good cycling ability we mean the charge acceptance after deep discharge is very good over repeated cycles, which is ideally suited for gulf conditions because of the specific driving practice and life style. Low maintenance means that the batteries will not require frequent topping up of water as compared to conventional automotive batteries. The topping frequency will be reduced to 40% in Hybrid Technology batteries as compared to conventional Automotive battery.
Sparco DS offers Batteries with superior performance and longer lifespan. It provides a complete range of VRLA & Lead Acid 2W Batteries. These Batteries are manufactured on a state of the art fully automatic assembly line with advanced technology, ensuring optimum performance, long life & reliable operation.
We offer Batteries with superior performance and longer lifespan. It provides a complete range of VRLA & Lead Acid 2W Batteries. These Batteries are manufactured on a state of the art fully automatic assembly line with advanced technology, ensuring optimum performance, long life & reliable operation.
Features-